We constructed an H5N1 AIV–chicken protein interaction map, and in addition to replicating previously identified host factors, we uncovered several novel interactions. Here, we applied the AP-MS technology to uncover a wide array of host proteins, complexes, and pathways that are hijacked by H5N1 avian influenza virus (AIV) during the course of infection. A comprehensive understanding of host–virus interactions would greatly improve our understanding of the viral life cycle and host resistance mechanisms. However, knowledge of common and strain-specific interactions remains incomplete, and how these interactions control host defense and viral infection remains to be fully elucidated. Several proteomic studies have used affinity purification mass spectrometry approaches to identify a series of cellular factors that interact with IAV proteins ( 8– 10). Viruses rely on host cellular functions to replicate, and thus, they hijack the host cell machinery and rewire it for their own needs. Upon infection by influenza, the host cells detect the viral RNA through pathogen sensors, and the major gene products of the influenza virus mediate the viral life cycle and modulate cellular processes ( 7). The vRNPs are surrounded by a layer of the matrix protein, M1, which lines the envelope ( 6). Inside the virion, all eight viral RNA (vRNA) segments bind three RNA polymerases (polymerase acid protein, PA polymerase basic protein 1, PB1 and 2, PB2) and are encapsidated by the nucleoprotein (NP) to form the viral ribonucleoprotein (vRNP) complexes ( 1– 5). IAV particles have two viral surface glycoproteins (hemagglutinin, HA neuraminidase, NA) and one matrix-2 protein (M2). This virus causes annual epidemics and recurring pandemics, which have huge impacts on public health. Influenza A virus (IAV) is a segmented, single-stranded, negative-sense RNA virus that has adapted to infect multiple species. This dataset facilitates a more comprehensive and detailed understanding of how the host machinery is manipulated during the course of H5N1 AIV infection. Specifically, we revealed that chicken Staufen double-stranded RNA-binding protein 2 interacts with AIV non-structural protein 1 (NS1) and promotes the replication of the virus by enhancing the nuclear export of NS1 mRNA. We identified with high confidence 1,043 H5N1 AIV–chicken interactions involving 621 individual chicken proteins and uncovered a number of host proteins and complexes that were targeted by the viral proteins. Here, we utilized affinity purification mass spectrometry (AP-MS) to systematically determine the physical interactions of 11 H5N1 AIV proteins with host proteins in chicken DF1 cells. To develop a comprehensive understanding of how H5N1 AIV rewires host cellular machinery during the course of infection, it is crucial to identify which host proteins and complexes come into physical contact with the viral proteins. 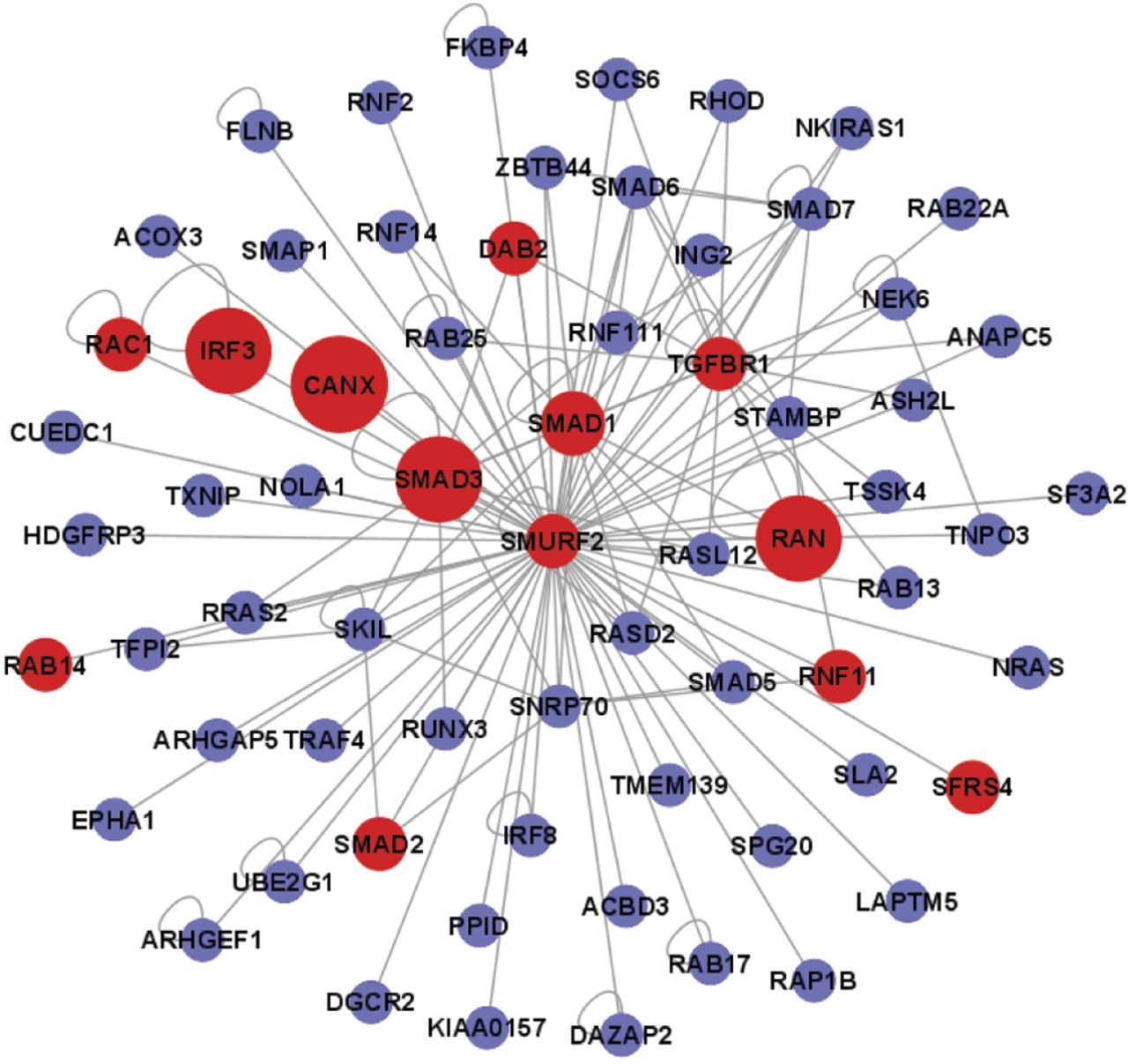
H5N1 AIV has a small genome and, therefore, relies heavily on its host cellular machinery to replicate. State Key Laboratory of Animal Nutrition, Institute of Animal Sciences, Chinese Academy of Agricultural Sciences, Beijing, ChinaĪs a highly pathogenic influenza virus, H5N1 avian influenza virus (AIV) poses a great threat to poultry production and public health.Qiao Wang †, Qi Zhang †, Maiqing Zheng, Jie Wen, Qinghe Li * and Guiping Zhao *
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
